This water tester lets you actually see what is dissolved in your drinking water!

This TDS Drinking water comparison tester- ELECTROLYZER tests for Total Dissolved Solids - $35...(please click on the ebay button above if you wish to purchase one of these) or email me and I will send you an invoice to pay via direct deposit.
A simple and very effective way to compare clean/dirty water at the same time. A picture is worth a 1000 words, so you can actually see the contaminants in your drinking water. Don’t waste time and money with technical gadgets that make water testing very complex and ambiguous. Just put two metal probes into one glass of tap water and the other two metal probes into either rain, distilled or bore water. Turn on the red light switch and you will see the difference in water quality in just 30 - 60 seconds.
Note: This electrolyzer has a safety fuse to prevent harm to the operator or electrolyzer, if you should forget and leave it on too long. This electrolyzer was originally designed for the US market but has been made available in Australia now with the use of a plug adapter that is supplied with it.
Colours formed in water due to electrolysis, from least TDS to most TDS…
Pale Yellow: Very clean water
Yellow: Separated Mineral Grains
Red & Yellow: Iron Rust
Blue: Organic Phosphorus Compound
Blackish Green: Copper Rust, Lead, Mercury etc.
Pitch Black: Much heavy metals – electrolyzer test is at its limit
FYI
How does an electrolyzer work? An electrolyzer is a piece of electrochemical apparatus (something that uses electricity and chemistry at the same time) designed to perform electrolysis: splitting a solution into the atoms from which it's made by passing electricity through it. Electrolysis was pioneered in the 18th century by British chemist Sir Humphry Davy (1778–1829), who used a primitive battery called a Voltaic pile to discover a number of chemical elements including sodium and potassium.An electrolyzer is a bit like a battery working in reverse:
- In a battery, you have chemicals packed into a sealed container with two electrical terminals dipping into them. When you connect the terminals into a circuit, the chemicals undergo reactions inside the container and produce electricity that flows through the circuit. (Read more about this in our main article on batteries.)
- In an electrolyzer, you place a solution in a container and dip two terminals into it. You connect the terminals up to a battery or other power supply and pass electricity through the solution. Chemical reactions take place and the solution splits up into its atoms. If the solution you use is pure water (H2O), you find it quickly splitting up into hydrogen gas (at the negative electrode) and oxygen gas (at the positive electrode). It's relatively easy to collect and store these gases for use in future.
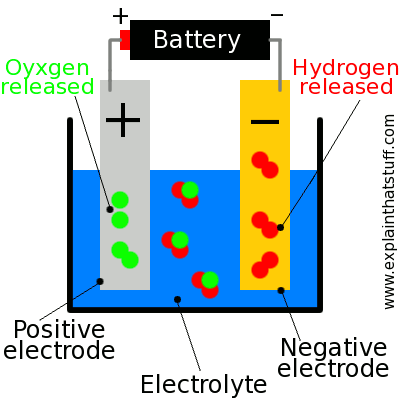
Here's how a simple electrolyzer makes hydrogen gas from water: (see photo at bottom)
{C}1. A battery connects the positive terminal (sometimes called the anode) to the negative terminal (or cathode) through an electrolyte. In a simple laboratory experiment, the electrolyte could be pure water. In a real electrolyzer, performance is improved considerably by using a solid polymer membrane as the electrolyte, which allows ions to move through it.
{C}2. When the power is switched on, water (H2O—shown here as two red blobs joined to one green one) splits into positively charged hydrogen ions (hydrogen atoms missing electrons, shown in red) and negatively charged oxygen ions (oxygen atoms with extra electrons, shown in green).
{C}3. The positive hydrogen ions are attracted to the negative terminal and recombine in pairs to form hydrogen gas (H2).
{C}4. Likewise, the negative oxygen ions are drawn to the positive terminal and recombine in pairs there to form oxygen gas (O2).
Q: Is an electrolysis water test accurate enough?
A: A test is conducted to help a certain decision, and the question whether or not it is accurate enough can only be examined in the context of the decision it was supposed to aid. To reject the electrolysis test because it is not as accurate as a full-fledged chemical analysis, is like rejecting an ECG (scan) because it is not as accurate as an Angiograph. Of course it is not. But if it answers the particular doubt the physician has, then it is “good enough”. In some cases angiography would be required. But we wouldn’t think much of a physician who sends every patient for an Angiograph, would we?
Thus, this simple electrolysis test presents the same facts to a person as an electronic TDS tester, but in a manner they can appreciate better. It is just like presenting some data as a pie-chart rather than as a table. In fact a table giving the exact values provides more information. But a pie-chart makes it easier to “grasp” that information. Likewise, an electrolysis test makes it easier for the average person to “grasp” the difference in water purity between two different sources.
Note: If you wish to purchase one of these electrolyzers then please click on the button at the end of this page.
Proudly powered by Weebly